Beyond compliance: How external quality assessment drives laboratory improvement over time
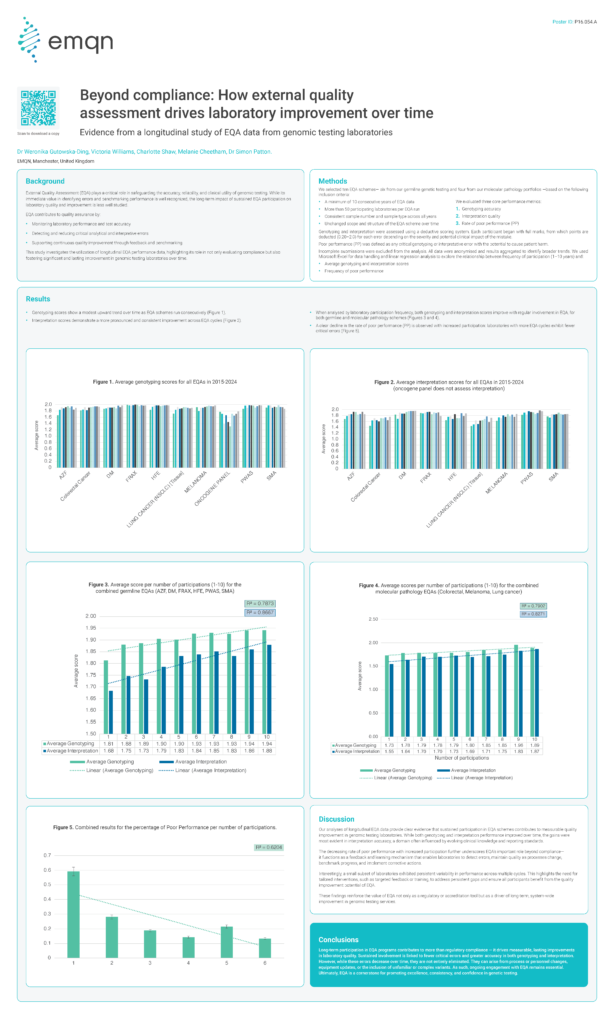
External Quality Assessment (EQA) plays a critical role in safeguarding the accuracy, reliability, and clinical utility of genomic testing. While its immediate value in identifying errors and benchmarking performance is well recognized, the long-term impact of sustained EQA participation on laboratory quality and improvement is less well studied. EQA contributes to quality assurance by:• Monitoring […]
Learnings from five years of External Quality Assessment (EQA) for porphyrias
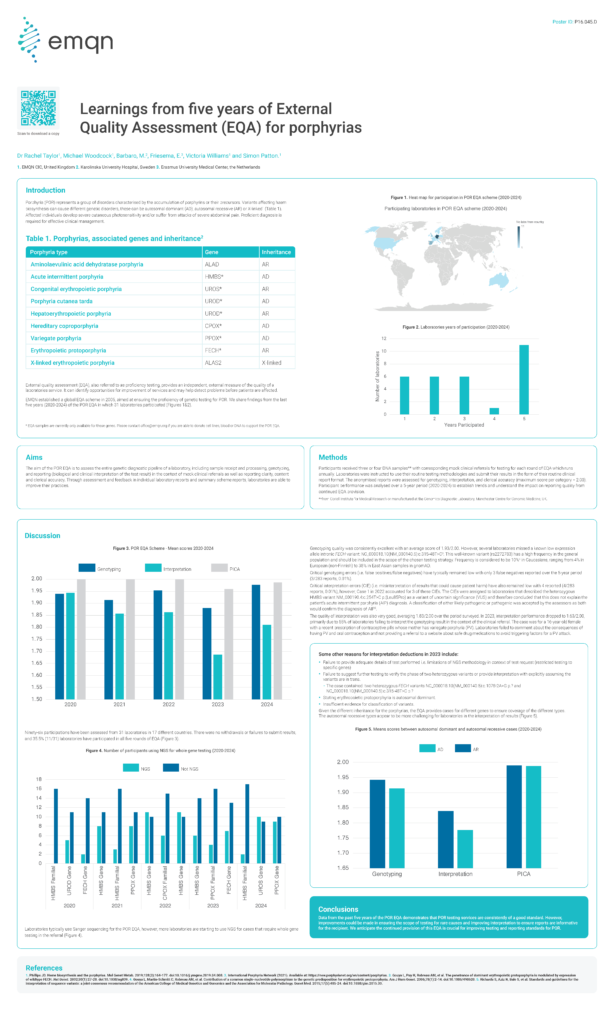
The aim of the POR EQA is to assess the entire genetic diagnostic pipeline of a laboratory, including sample receipt and processing, genotyping,and reporting (biological and clinical interpretation of the test result) in the context of mock clinical referrals as well as reporting clarity, contentand clerical accuracy. Through assessment and feedback in individual laboratory reports […]
Common causes of genotyping error; Trends over time
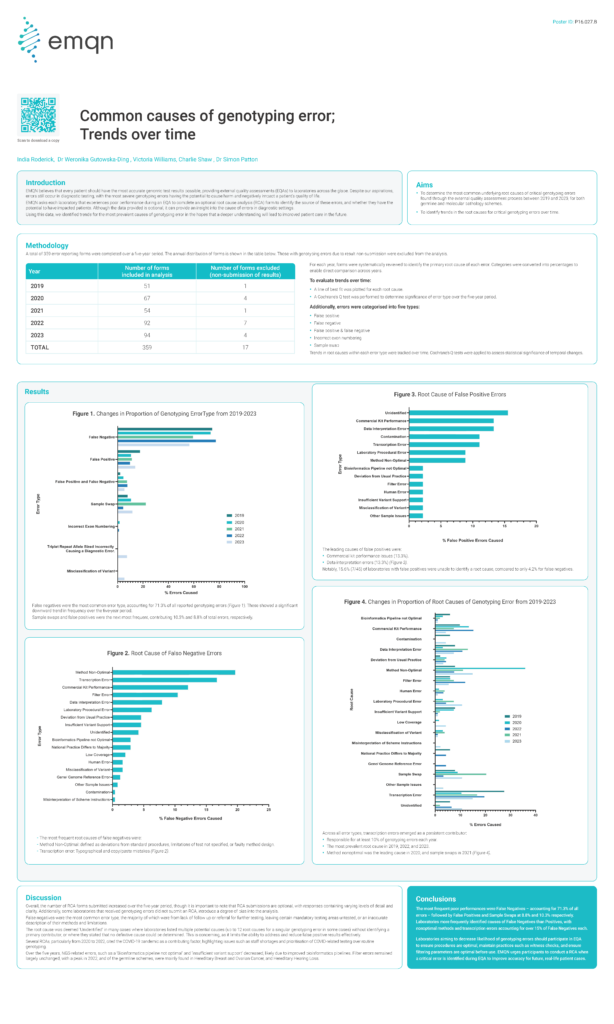
EMQN believes that every patient should have the most accurate genomic test results possible, providing external quality assessments (EQAs) to laboratories across the globe. Despite our aspirations,errors still occur in diagnostic testing, with the most severe genotyping errors having the potential to cause harm and negatively impact a patient’s quality of life.EMQN asks each laboratory […]
External Quality Assessment for hereditary amyloidosis: Assuring test standards to support the identification of patients eligible for treatment

Hereditary amyloidosis is a group of rare autosomal dominant conditions characterised by amyloid accumulation impairing organ function. Hereditary transthyretin amyloidosis (ATTRv) – the most common type – is caused by >130 pathogenic variants in the TTR gene and presents with neurological and/or cardiac manifestations. Several disease modifying therapies for ATTRv are now available, making a […]
External Quality Assessment (EQA) for Homologous Recombination Deficiency (HRD) testing in ovarian cancer: Findings of a new international Quality Network for Pathology (IQN Path) pilot scheme

HRD occurs when cells lose their ability to repair double-stranded DNA breaks via the high-fidelity, homologous recombination repair (HRR) mechanism. Multiple genes mediate the HRR pathway with BRCA1/BRCA2 being the most well characterized. However, HRD can also be a consequence of defects in other HRR genes and may lead to genomic structural alterations known as […]
Learnings from a Pilot External Quality Assessment (EQA) for cfDNA Extraction from Plasma
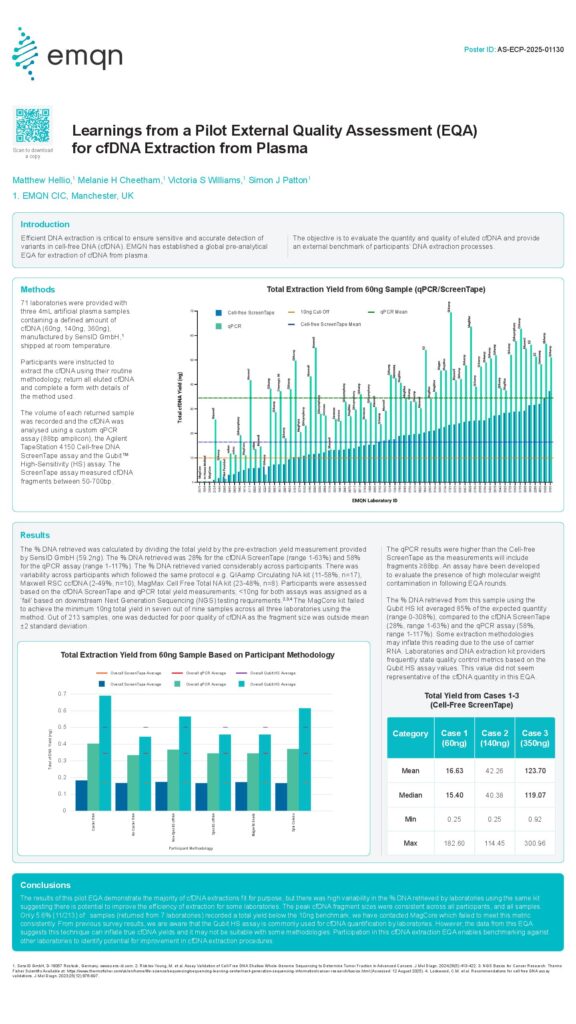
Efficient DNA extraction is critical to ensure sensitive and accurate detection of variants in cell-free DNA (cfDNA). EMQN has established a global pre-analytical EQA for extraction of cfDNA from plasma. The objective is to evaluate the quantity and quality of eluted cfDNA and provide an external benchmark of participants‘ DNA extraction processes.
