Beyond compliance: How external quality assessment drives laboratory improvement over time
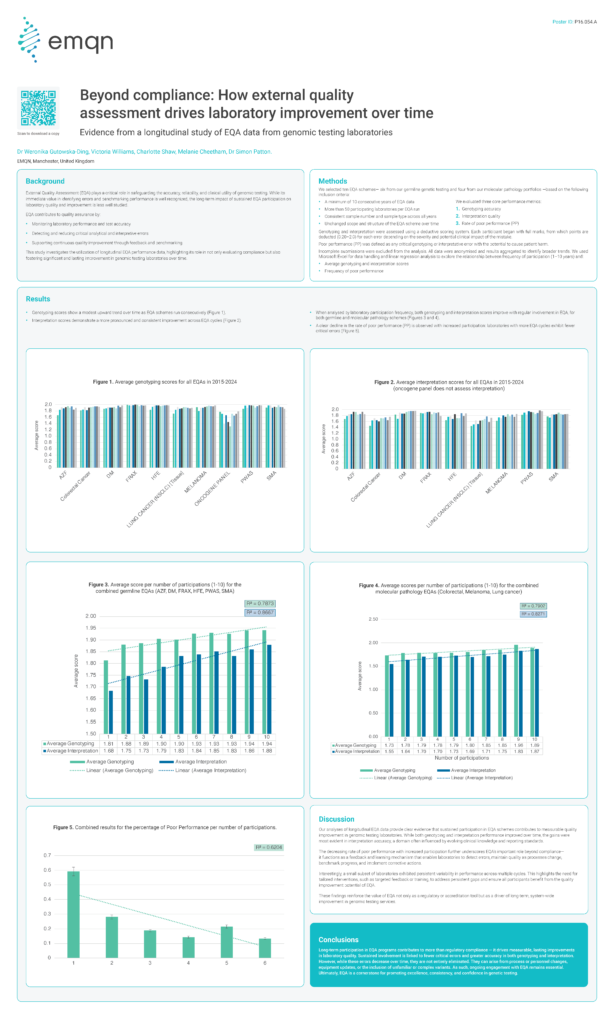
External Quality Assessment (EQA) plays a critical role in safeguarding the accuracy, reliability, and clinical utility of genomic testing. While its immediate value in identifying errors and benchmarking performance is well recognized, the long-term impact of sustained EQA participation on laboratory quality and improvement is less well studied. EQA contributes to quality assurance by:• Monitoring […]
External Quality Assessment (EQA) for Homologous Recombination Deficiency (HRD) testing in ovarian cancer: Findings of a new international Quality Network for Pathology (IQN Path) pilot scheme

HRD occurs when cells lose their ability to repair double-stranded DNA breaks via the high-fidelity, homologous recombination repair (HRR) mechanism. Multiple genes mediate the HRR pathway with BRCA1/BRCA2 being the most well characterized. However, HRD can also be a consequence of defects in other HRR genes and may lead to genomic structural alterations known as […]
Learnings from a Pilot External Quality Assessment (EQA) for cfDNA Extraction from Plasma
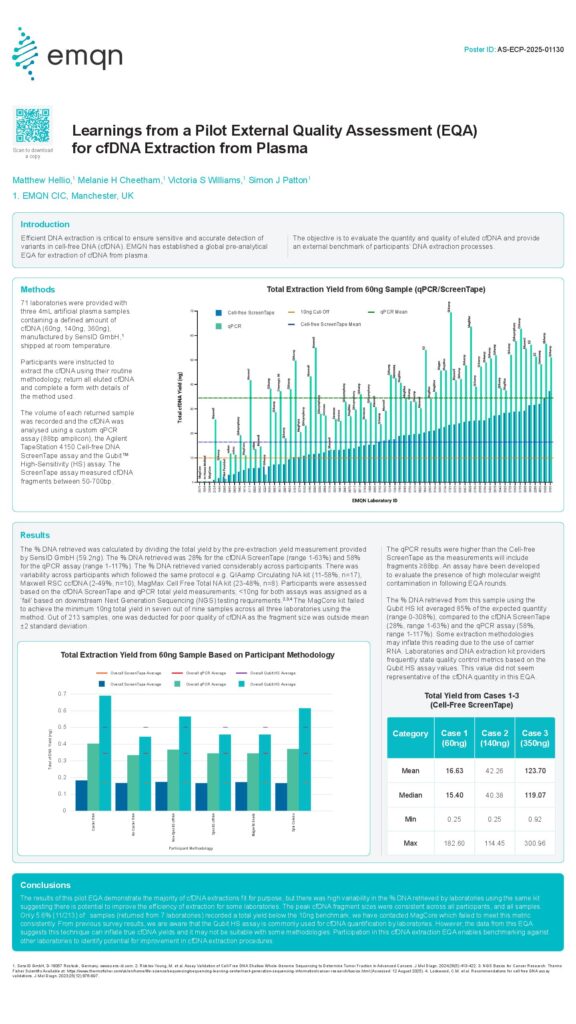
Efficient DNA extraction is critical to ensure sensitive and accurate detection of variants in cell-free DNA (cfDNA). EMQN has established a global pre-analytical EQA for extraction of cfDNA from plasma. The objective is to evaluate the quantity and quality of eluted cfDNA and provide an external benchmark of participants‘ DNA extraction processes.
